For a complex number $z$, let $Re(z)$ denote the real part of $z$. Let $S$ be the set of all complex numbers $z$ satisfying $z^{4}-|z|^{4}=4 i z^{2}$, where $i=\sqrt{-1}$. Then the minimum possible value of $\left|z_{1}-z_{2}\right|^{2}$, where $z_{1}, z_{2} \in S$ with \(Re\left( z _{1}\right)\)\(>\)0 and \(Re\left( z _{2}\right)\) \(<\) 0, is ______
Correct Answer: 8
Solution and Explanation
\(|z_1 – z_2|^ 2 = 8\)
Top Questions on Complex numbers
- A building construction work can be completed by two masons A and B together in 22.5 days. Mason A alone can complete the construction work in 24 days less than mason B alone. Then mason A alone will complete the construction work in :
- JEE Main - 2026
- Mathematics
- Complex numbers
- Let \( z \) be a complex number such that \( |z-6|=5 \) and \( |z+2-6i|=5 \). Then the value of \( z^3+3z^2-15z+14 \) is equal to:
- JEE Main - 2026
- Mathematics
- Complex numbers
Let \(S=\left\{ z\in\mathbb{C}:\left|\frac{z-6i}{z-2i}\right|=1 \text{ and } \left|\frac{z-8+2i}{z+2i}\right|=\frac{3}{5} \right\}.\)
Then $\sum_{z\in S}|z|^2$ is equal to
- JEE Main - 2026
- Mathematics
- Complex numbers
- If \(x^2+x+1=0\), then the value of \(\left(x + \frac{1}{x}\right)^2 + \left(x^2 + \frac{1}{x^2}\right)^2 + \left(x^3 + \frac{1}{x^3}\right)^2 + \dots + \left(x^{25} + \frac{1}{x^{25}}\right)^2\) is:
- JEE Main - 2026
- Mathematics
- Complex numbers
- Let \[ S = \{ z \in \mathbb{C} : 4z^2 + \bar{z} = 0 \}. \] Then \[ \sum_{z \in S} |z|^2 \] is equal to
- JEE Main - 2026
- Mathematics
- Complex numbers
Questions Asked in JEE Advanced exam
- Let $ x_0 $ be the real number such that $ e^{x_0} + x_0 = 0 $. For a given real number $ \alpha $, define $$ g(x) = \frac{3xe^x + 3x - \alpha e^x - \alpha x}{3(e^x + 1)} $$ for all real numbers $ x $. Then which one of the following statements is TRUE?
- JEE Advanced - 2025
- Fundamental Theorem of Calculus
- A linear octasaccharide (molar mass = 1024 g mol$^{-1}$) on complete hydrolysis produces three monosaccharides: ribose, 2-deoxyribose and glucose. The amount of 2-deoxyribose formed is 58.26 % (w/w) of the total amount of the monosaccharides produced in the hydrolyzed products. The number of ribose unit(s) present in one molecule of octasaccharide is _____.
Use: Molar mass (in g mol$^{-1}$): ribose = 150, 2-deoxyribose = 134, glucose = 180; Atomic mass (in amu): H = 1, O = 16- JEE Advanced - 2025
- Biomolecules
Let $ P(x_1, y_1) $ and $ Q(x_2, y_2) $ be two distinct points on the ellipse $$ \frac{x^2}{9} + \frac{y^2}{4} = 1 $$ such that $ y_1 > 0 $, and $ y_2 > 0 $. Let $ C $ denote the circle $ x^2 + y^2 = 9 $, and $ M $ be the point $ (3, 0) $. Suppose the line $ x = x_1 $ intersects $ C $ at $ R $, and the line $ x = x_2 $ intersects $ C $ at $ S $, such that the $ y $-coordinates of $ R $ and $ S $ are positive. Let $ \angle ROM = \frac{\pi}{6} $ and $ \angle SOM = \frac{\pi}{3} $, where $ O $ denotes the origin $ (0, 0) $. Let $ |XY| $ denote the length of the line segment $ XY $. Then which of the following statements is (are) TRUE?
- JEE Advanced - 2025
- Conic sections
- Adsorption of phenol from its aqueous solution on to fly ash obeys Freundlich isotherm. At a given temperature, from 10 mg g$^{-1}$ and 16 mg g$^{-1}$ aqueous phenol solutions, the concentrations of adsorbed phenol are measured to be 4 mg g$^{-1}$ and 10 mg g$^{-1}$, respectively. At this temperature, the concentration (in mg g$^{-1}$) of adsorbed phenol from 20 mg g$^{-1}$ aqueous solution of phenol will be ____. Use: $\log_{10} 2 = 0.3$
- JEE Advanced - 2025
- Adsorption
- At 300 K, an ideal dilute solution of a macromolecule exerts osmotic pressure that is expressed in terms of the height (h) of the solution (density = 1.00 g cm$^{-3}$) where h is equal to 2.00 cm. If the concentration of the dilute solution of the macromolecule is 2.00 g dm$^{-3}$, the molar mass of the macromolecule is calculated to be $X \times 10^{4}$ g mol$^{-1}$. The value of $X$ is ____. Use: Universal gas constant (R) = 8.3 J K$^{-1}$ mol$^{-1}$ and acceleration due to gravity (g) = 10 m s$^{-2}\}$
- JEE Advanced - 2025
- Colligative Properties
Concepts Used:
Law of Chemical Equilibrium
Law of Chemical Equilibrium states that at a constant temperature, the rate of a chemical reaction is directly proportional to the product of the molar concentrations of the reactants each raised to a power equal to the corresponding stoichiometric coefficients as represented by the balanced chemical equation.
Let us consider a general reversible reaction;
A+B ↔ C+D
After some time, there is a reduction in reactants A and B and an accumulation of the products C and D. As a result, the rate of the forward reaction decreases and that of backward reaction increases.
Eventually, the two reactions occur at the same rate and a state of equilibrium is attained.
By applying the Law of Mass Action;
The rate of forward reaction;
Rf = Kf [A]a [B]b
The rate of backward reaction;
Rb = Kb [C]c [D]d
Where,
[A], [B], [C] and [D] are the concentrations of A, B, C and D at equilibrium respectively.
a, b, c, and d are the stoichiometric coefficients of A, B, C and D respectively.
Kf and Kb are the rate constants of forward and backward reactions.
However, at equilibrium,
Rate of forward reaction = Rate of backward reaction.
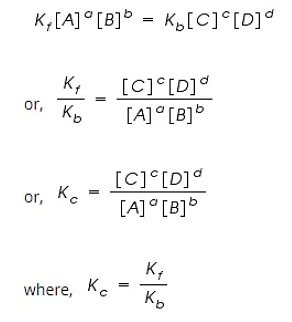
Kc is called the equilibrium constant expressed in terms of molar concentrations.
The above equation is known as the equation of Law of Chemical Equilibrium.