The pH of an aqueous buffer prepared using CH3COOH and CH3COO Na+ is 4.80.
The quantity \(\frac{[CH_3COO^-]-[CH_3COOH]}{[CH_3COOH]}\) is _____. (round off to three decimal places)
[Given: pKa of CH3COOH in water is 4.75]
The quantity \(\frac{[CH_3COO^-]-[CH_3COOH]}{[CH_3COOH]}\) is _____. (round off to three decimal places)
[Given: pKa of CH3COOH in water is 4.75]
Correct Answer: 0.121
Solution and Explanation
To solve for \(\frac{[CH_3COO^-]-[CH_3COOH]}{[CH_3COOH]}\), we use the Henderson-Hasselbalch equation:
\[\text{pH} = \text{p}K_a + \log\left(\frac{[CH_3COO^-]}{[CH_3COOH]}\right)\]
Given:
pH = 4.80
pK\(_a\) of CH\(_3\)COOH = 4.75
Substitute the values:
4.80 = 4.75 + \log\left(\frac{[CH_3COO^-]}{[CH_3COOH]}\right)
Solve for \(\log\left(\frac{[CH_3COO^-]}{[CH_3COOH]}\right)\):
\[\log\left(\frac{[CH_3COO^-]}{[CH_3COOH]}\right) = 4.80 - 4.75 = 0.05\]
Convert the logarithm to a ratio:
\[\frac{[CH_3COO^-]}{[CH_3COOH]} = 10^{0.05}\]
Calculate \(10^{0.05}\):
\(10^{0.05} \approx 1.122\)
Thus, \(\frac{[CH_3COO^-]-[CH_3COOH]}{[CH_3COOH]}\) can be determined:
\(\frac{[CH_3COO^-]-[CH_3COOH]}{[CH_3COOH]} = \left(\frac{[CH_3COO^-]}{[CH_3COOH]}-1\right)\)
Substitute the calculated ratio:
\(\frac{[CH_3COO^-]-[CH_3COOH]}{[CH_3COOH]} = 1.122 - 1 = 0.122\)
Round to three decimal places: **0.122**
Top Questions on Chemical equilibria
- The ratio of osmotic pressures of aqueous solutions of 0.01 M BaCl2 to 0.005 M NaCl is
[Given: Both compounds dissociate completely in water]- IIT JAM CY - 2024
- Physical Chemistry
- Chemical equilibria
- A 1.0 L solution is prepared by dissolving 2.0 g of benzoic acid and 4.0 g of sodium benzoate in water. The pH of the resulting solution is _______. (rounded off to one decimal place)
Given: Molar mass of benzoic acid is 122 g mol−1
Molar mass of sodium benzoate is 144 g mol−1
p𝐾a of benzoic acid is 4.2- IIT JAM CY - 2024
- Physical Chemistry
- Chemical equilibria
- 0.1 M aqueous solution of a weak monobasic acid has pH 2.0. The pKa of the monobasic acid is _______. (rounded off to one decimal place)
- IIT JAM CY - 2024
- Physical Chemistry
- Chemical equilibria
- Consider the exothermic chemical reaction O2(𝑔)+2H2(𝑔) ⇌ 2H2O(𝑔) at equilibrium in a closed container. The correct statement(s) is/are
- IIT JAM CY - 2024
- Physical Chemistry
- Chemical equilibria
- The isoelectric point of glutamic acid is ______.
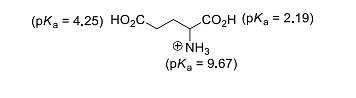
(round off to two decimal places)- IIT JAM CY - 2023
- Physical Chemistry
- Chemical equilibria
Questions Asked in IIT JAM CY exam
- Among the following, the correct condition(s) for spontaneity is(are)
- IIT JAM CY - 2025
- Thermodynamics
One mole of a monoatomic ideal gas starting from state A, goes through B and C to state D, as shown in the figure. Total change in entropy (in J K\(^{-1}\)) during this process is ...............

- IIT JAM CY - 2025
- Thermodynamics
The number of chiral carbon centers in the following molecule is ...............

- IIT JAM CY - 2025
- General Chemistry
- Consider the following matrices A and B.
\[ A = \begin{pmatrix} 1 & 2 & 0 & 0 \\ 3 & 4 & 0 & 0 \\ 0 & 5 & 5 & 0 \\ 0 & 0 & 6 & 7 \\ 0 & 0 & 8 & 9 \end{pmatrix} \quad \text{and} \quad B = \begin{pmatrix} 10 & 11 & 0 & 0 & 0 \\ 12 & 13 & 0 & 0 & 0 \\ 0 & 0 & 4 & 0 & 0 \\ 0 & 0 & 15 & 16 & 0 \\ 0 & 0 & 17 & 18 & 0 \end{pmatrix} \]
If \( C = AB \), the sum of the diagonal elements of \( C \) is ..............
- IIT JAM CY - 2025
- General Chemistry
A tube fitted with a semipermeable membrane is dipped into 0.001 M NaCl solution at 300 K as shown in the figure. Assume density of the solvent and solution are the same. At equilibrium, the height of the liquid column \( h \) (in cm) is .........

- IIT JAM CY - 2025
- General Chemistry