Question:
When limestone is heated, $CO_2$ is given off. The metallurgical operation is
When limestone is heated, $CO_2$ is given off. The metallurgical operation is
Updated On: Apr 2, 2024
- Smelting
- Reduction
- Calcination
- Roasting
Hide Solution
Verified By Collegedunia
The Correct Option is C
Solution and Explanation
Calcination is the process of converting an ore into its oxide by heating it strongly below its melting point either in absence or limited supply of air. Carbonate ores are converted into their respective oxides by loss of carbon dioxide.
$CaCO _{3} {->[{\Delta}]} \underset {\text {Calcium oxide }}{CaO} + CO _{2} \uparrow$
$CaCO _{3} {->[{\Delta}]} \underset {\text {Calcium oxide }}{CaO} + CO _{2} \uparrow$
Was this answer helpful?
0
0
Top Questions on General Principles and Processes of Isolation of Elements
- What is used for the Thermite Reaction?
- BCECE - 2025
- Chemistry
- General Principles and Processes of Isolation of Elements
- In the extraction of iron using blast furnace to remove the impurity (X), chemical (Y) is added to the ore. X and Y are respectively
- TS EAMCET - 2025
- Chemistry
- General Principles and Processes of Isolation of Elements
- Which of the following compounds is used to cover the surface of a metallic object to prevent corrosion?
- KEAM - 2025
- Chemistry
- General Principles and Processes of Isolation of Elements
- The incorrect statement about the Hall-Heroult process is:
- KCET - 2024
- Chemistry
- General Principles and Processes of Isolation of Elements
- Select the correct statement:
- KCET - 2024
- Chemistry
- General Principles and Processes of Isolation of Elements
View More Questions
Questions Asked in KCET exam
Match the following:
In the following, \( [x] \) denotes the greatest integer less than or equal to \( x \).
Choose the correct answer from the options given below:- KCET - 2025
- Differentiability
- If \[ y = \frac{\cos x}{1 + \sin x} \] then:
- KCET - 2025
- Differentiability
- A function \( f(x) \) is given by:
\[ f(x) = \begin{cases} \frac{1}{e^x - 1}, & \text{if } x \neq 0 \\ \frac{1}{e^x + 1}, & \text{if } x = 0 \end{cases} \] Then, which of the following is true?- KCET - 2025
- Limits
- The function f(x) is given by:
For x < 0:
f(x) = ex + axFor x ≥ 0:
f(x) = b(x - 1)2
The function is differentiable at x = 0. Then,- KCET - 2025
- Differentiability
- The function \( f(x) = \tan x - x \)
- KCET - 2025
- Derivatives
View More Questions
Concepts Used:
General Principles and Processes of Isolation of Elements
What are Ores and Minerals?
Minerals are the naturally occurring, homogeneous inorganic solid substances. They are having a definite chemical composition and crystalline structure, hardness and color. For example, copper pyrite, calamine, etc.
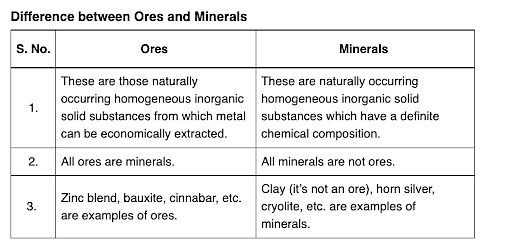
Impurities in an ore are called gauge. The removal of a gauge from the ore is called concentration ore.
Several steps are involved in the extraction of pure metal from ores. Major steps are as follows –
- Concentration of the ore
- Isolation of the metal from its concentrated ore
- Purification of the metal