Question:
The number of zinc ions present in the molecular formula of \( Q \) is:
The number of zinc ions present in the molecular formula of \( Q \) is:
Show Hint
Consider both stoichiometric possibilities for product formation.
Updated On: Jan 20, 2025
Hide Solution
Verified By Collegedunia
Solution and Explanation
Step 1: Analyze the reaction pathways.
\[
K_4[Fe(CN)_6] + 3ZnCl_2 \rightarrow K_2Zn_3[Fe(CN)_6]_2 + 6KCl
\]
This gives 3 zinc ions.
Alternatively:
\[
K_4[Fe(CN)_6] + 2ZnCl_2 \rightarrow Zn_2[Fe(CN)_6] + 4KCl
\]
This gives 2 zinc ions.
Was this answer helpful?
0
0
Top Questions on Qualitative Analysis
- Given below are two statements:
Statement I: In Lassaigne's test, the covalent organic molecules are transformed into ionic compounds.
Statement II: The sodium fusion extract of an organic compound having N and S gives prussian blue colour with FeSO4 and Na4[Fe(CN)6].
In the light of the above statements, choose the correct answer from the options given below.- JEE Main - 2025
- Chemistry
- Qualitative Analysis
Among the following cations, the number of cations which will give characteristic precipitate in their identification tests with
\(K_4\)[Fe(CN)\(_6\)] is : \[ {Cu}^{2+}, \, {Fe}^{3+}, \, {Ba}^{2+}, \, {Ca}^{2+}, \, {NH}_4^+, \, {Mg}^{2+}, \, {Zn}^{2+} \]- JEE Main - 2025
- Chemistry
- Qualitative Analysis
- An organic compound \(P\) with molecular formula \(C_6H_6O_3\) gives a ferric chloride test and does not have an intramolecular hydrogen bond. The compound \(P\) reacts with 3 equivalents of \(NH_2OH\) to produce oxime \(Q\). Treatment of \(P\) with excess methyl iodide in the presence of \(KOH\) produces compound \(R\) as the major product. Reaction of \(R\) with excess iso-butylmagnesium bromide followed by treatment with \(H_3O^+\) gives compound \(S\) as the major product. The total number of methyl (\(-CH_3\)) groups in compound \(S\) is \_\_\_\_\_.
- JEE Advanced - 2024
- Chemistry
- Qualitative Analysis
- Match List-I with List-II :Identify the correct answer from the options given below :
List - I(Test/reagent) List - II(Radical identified) (A) Lake Test (I) NO3− (B) Nessler’s Reagent (II) Fe3+ (C) Potassium sulphocyanide (III) Al3+ (D) Brown Ring Test (IV) NH4+ - NEET (UG) - 2024
- Chemistry
- Qualitative Analysis
- Match List-I with List-II :Identify the correct answer from the options given below :
List - ISolid salt treated with dil. H2SO4 List - IIAnion detected (A) effervescence of colourless gas (I) NO2− (B) gas with smell of rotten egg (II) CO32− (C) gas with pungent smell (III) S2− (D) brown fumes (IV) SO23− - NEET (UG) - 2024
- Chemistry
- Qualitative Analysis
View More Questions
Questions Asked in JEE Advanced exam
- A region in the form of an equilateral triangle (in x-y plane) of height L has a uniform magnetic field 𝐵⃗ pointing in the +z-direction. A conducting loop PQR, in the form of an equilateral triangle of the same height 𝐿, is placed in the x-y plane with its vertex P at x = 0 in the orientation shown in the figure. At 𝑡 = 0, the loop starts entering the region of the magnetic field with a uniform velocity 𝑣 along the +x-direction. The plane of the loop and its orientation remain unchanged throughout its motion.
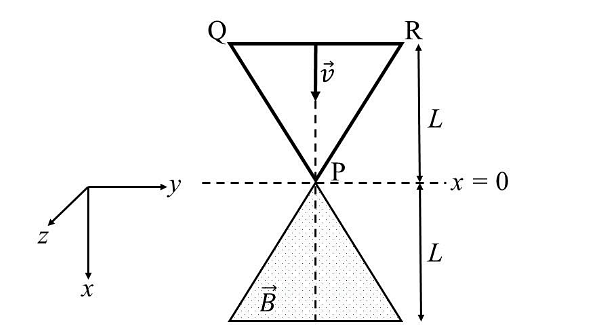
Which of the following graph best depicts the variation of the induced emf (E) in the loop as a function of the distance (𝑥) starting from 𝑥 = 0? - A positive, singly ionized atom of mass number \(A_M\) is accelerated from rest by the voltage \(192 V\). Thereafter, it enters a rectangular region of width 𝑤 with magnetic field \(\bar{B_0} = 0.1𝑘̂\)Tesla, as shown in the figure. The ion finally hits a detector at the distance 𝑥 below its starting trajectory. [Given: Mass of neutron/proton =\((5/3) × 10^{−27}\) kg, charge of the electron \(= 1.6 × 10^{−19} C.\)]
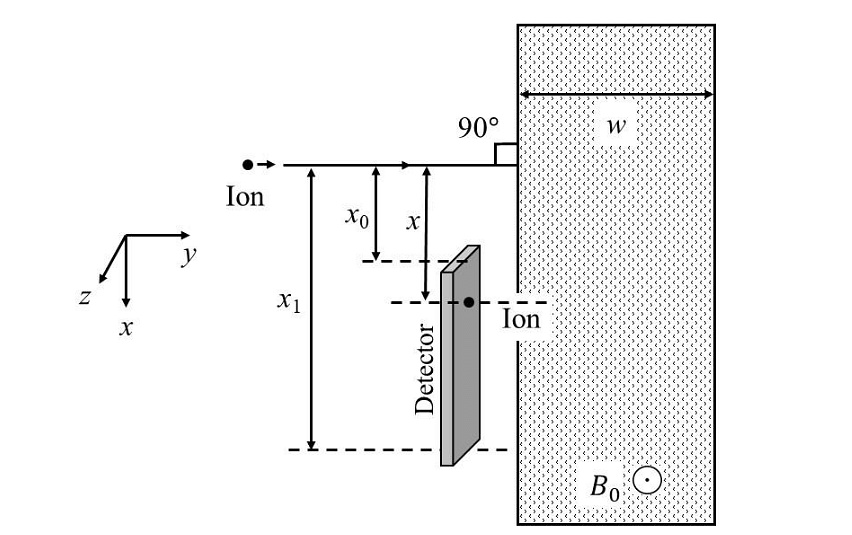
Which of the following option(s) is(are) correct?- JEE Advanced - 2024
- Electromagnetic waves
- Among the following, the correct statement(s) for electrons in an atom is(are)
- JEE Advanced - 2024
- Structure of atom
- An organic compound P having molecular formula C6H6O3 gives ferric chloride test and does not have intramolecular hydrogen bond. The compound P reacts with 3 equivalents of NH2OH to produce oxime Q. Treatment of P with excess methyl iodide in the presence of KOH produces compound R as the major product. Reaction of R with excess iso-butylmagnesium bromide followed by treatment with H3O+ gives compound S as the major product.
The total number of methyl (−CH3) group(s) in compound S is ____.- JEE Advanced - 2024
- Aldehydes, Ketones and Carboxylic Acids
- Among the following options, select the option in which each complex in Set-I shows geometrical isomerism and the two complexes in Set-II are ionization isomers of each other.
[en = H2NCH2CH2NH2]- JEE Advanced - 2024
- coordination compounds
View More Questions