Although the details of the structure of monoclinic sulphur are not well known it probably consists of
- $S_8$ chains
- $S_2$ molecule
- $S_8$rings
- $S_4$ rings
The Correct Option is C
Approach Solution - 1
Approach Solution -2
The structure of monoclinic sulfur is not perfectly understood, but it's widely accepted that monoclinic sulfur primarily consists of S8 rings.
The elemental form of sulfur (S) usually exists as S8 molecules, where eight sulfur atoms are arranged in a ring. These S8 rings can further form various structures. In monoclinic sulfur, these rings are thought to arrange themselves in such a way that they form long chains. This arrangement allows monoclinic sulfur to exhibit unique physical properties.
So, the correct option is (C): S8 rings
Top Questions on Group 16 Elements
- Why does oxygen show anomalous behavior ?
- JEE Main - 2024
- Chemistry
- Group 16 Elements
- Choose the correct statements from the following:
A. All group 16 elements form oxides of general formula \( EO_2 \) and \( EO_3 \) where \( E = S, Se, Te, \) and Po. Both the types of oxides are acidic in nature.
B. TeO\(_2\) is an oxidising agent while SO\(_2\) is reducing in nature.
C. The reducing property decreases from H\(_2\)S to H\(_2\)Te down the group.
D. The ozone molecule contains five lone pairs of electrons.
Choose the correct answer from the options given below:- JEE Main - 2024
- Chemistry
- Group 16 Elements
- From the given list, the number of compounds with +4 oxidation state of Sulphur_____.
\(SO_3, H_2SO_3, SOCl_2, SF_4, BaSO_4, H_2S_2O_7\).- JEE Main - 2024
- Chemistry
- Group 16 Elements
- Among Group 16 elements, which one does NOT show -2 oxidation state?
- NEET (UG) - 2024
- Chemistry
- Group 16 Elements
- Given below are two statements :
Statement I: The boiling point of hydrides of Group 16 elements follow the order
H2O > H2Te > H2Se > H2S.
Statement II: On the basis of molecular mass, H2O is expected to have lower boiling point than the other members of the group but due to the presence of extensive H-bonding in H2O, it has higher boiling point.
In the light of the above statements, choose the correct answer from the options given below :- NEET (UG) - 2024
- Chemistry
- Group 16 Elements
Questions Asked in CBSE CLASS XII exam
If vector \( \mathbf{a} = 3 \hat{i} + 2 \hat{j} - \hat{k} \) \text{ and } \( \mathbf{b} = \hat{i} - \hat{j} + \hat{k} \), then which of the following is correct?
- Find the value of $x$, if \[ \begin{bmatrix} 1 & 3 & 2 \\ 2 & 5 & 1 \\ 15 & 3 & 2 \end{bmatrix} \begin{bmatrix} 1 \\ x \\ 2 \end{bmatrix} = \begin{bmatrix} 0 \\ 0 \\ 0 \end{bmatrix} \]
- Two point charges of \( -5\,\mu C \) and \( 2\,\mu C \) are located in free space at \( (-4\,\text{cm}, 0) \) and \( (6\,\text{cm}, 0) \) respectively.
(a) Calculate the amount of work done to separate the two charges at infinite distance.
(b) If this system of charges was initially kept in an electric field \[ \vec{E} = \frac{A}{r^2}, \text{ where } A = 8 \times 10^4\, \text{N}\,\text{C}^{-1}\,\text{m}^2, \] calculate the electrostatic potential energy of the system.- CBSE CLASS XII - 2025
- Electrostatics
- 4,000 shares of ₹ 10 each were forfeited for non-payment of second and final call money of ₹ 2 per share. The minimum amount that the company must collect at the time of reissue of these shares will be :
- CBSE CLASS XII - 2025
- Accounting for Share Capital
- If $y = a \cos(\log x) + b \sin(\log x)$, then $x^2y'' + xy'1$ is:
- CBSE CLASS XII - 2025
- Continuity and differentiability
Concepts Used:
Group 16 Elements
The group 16 elements (oxygen group elements) of the periodic classification are also known as chalcogens because most of the copper ores have copper in the form of oxides and sulphides. The word chalcogen means “ore formation” which is derived from the Greek word “Chalcos” (Ore) and “gen” (formation).
There are 5 elements that come under Group 16 of the Modern Periodic Table namely:
- Oxygen (O)
- Sulphur (S)
- Selenium (Se)
- Tellurium (Te)
- Polonium (PO)
Electronic Configuration:
The general electronic configuration of the chalcogens can be written as ‘ns2np4’, where ‘n’ denotes the value of the principal quantum number corresponding to the valence shell of the element.
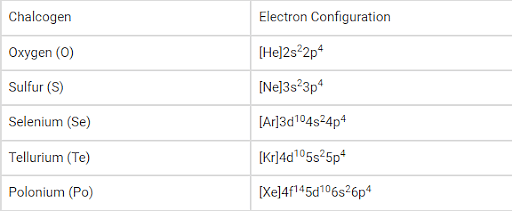
The electron configuration of the synthetic element livermorium (believed to be a chalcogen) is predicted to be [Rn]5f146d107s27p4.
Metallic Nature of the Group 16 Elements:
- Oxygen and sulfur are classified as non-metals.
- Selenium and tellurium are classified as metalloids.
- Under standard conditions, polonium exhibits metallic characteristics. However, it is important to note that polonium is a radioactive element.