Question:
For the following compound
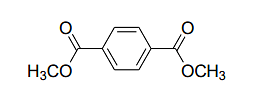
the number of signals expected in the 1H NMR spectrum is _________.
For the following compound

the number of signals expected in the 1H NMR spectrum is _________.

the number of signals expected in the 1H NMR spectrum is _________.
Updated On: Jan 11, 2025
Hide Solution
Verified By Collegedunia
Correct Answer: 2
Solution and Explanation
In the given compound, we have two distinct types of protons:
- The two methoxy groups (−OCH3) each have identical protons, and they are chemically equivalent.
- The protons on the benzene ring are all equivalent because of the symmetry of the structure (with methoxy groups at the ortho positions relative to each other).
Therefore, the 1H NMR spectrum will exhibit two signals:
- A signal for the methoxy protons (OCH3).
- A signal for the aromatic protons on the benzene ring.
Thus, the correct number of signals is 2
Was this answer helpful?
0
0
Top Questions on Spectroscopy
- In IR spectroscopy, which bond absorbs at the highest wavenumber?
- GATE CY - 2026
- Chemistry
- Spectroscopy
- Electromagnetic wave with intensity \( I = 4 \times 10^{14} \, \text{watt/m}^2 \) is propagating in free space. Find the amplitude of magnetic field \( B_0 \). Given: \( c = 3 \times 10^8 \, \text{m/s}, \epsilon_0 = 8.85 \times 10^{-12} \, \text{C}^2/\text{N} \cdot \text{m}^2 \).
- JEE Main - 2026
- Physics
- Spectroscopy
- . Number of different $\text{H}$-atoms in $(\text{Y})$

- JEE Main - 2026
- Chemistry
- Spectroscopy
- Among the following complex ions, the one which is EPR active is:
- OJEE - 2025
- Chemistry
- Spectroscopy
- The total number of hyperfine lines expected in the EPR spectrum of \(\cdot\)CHsubscript{2OH (radical) is ___________ (in integer).}
- GATE CY - 2025
- Chemistry
- Spectroscopy
View More Questions
Questions Asked in IIT JAM CY exam
- Among the following, the correct condition(s) for spontaneity is(are)
- IIT JAM CY - 2025
- Thermodynamics
One mole of a monoatomic ideal gas starting from state A, goes through B and C to state D, as shown in the figure. Total change in entropy (in J K\(^{-1}\)) during this process is ...............

- IIT JAM CY - 2025
- Thermodynamics
The number of chiral carbon centers in the following molecule is ...............

- IIT JAM CY - 2025
- General Chemistry
- Consider the following matrices A and B.
\[ A = \begin{pmatrix} 1 & 2 & 0 & 0 \\ 3 & 4 & 0 & 0 \\ 0 & 5 & 5 & 0 \\ 0 & 0 & 6 & 7 \\ 0 & 0 & 8 & 9 \end{pmatrix} \quad \text{and} \quad B = \begin{pmatrix} 10 & 11 & 0 & 0 & 0 \\ 12 & 13 & 0 & 0 & 0 \\ 0 & 0 & 4 & 0 & 0 \\ 0 & 0 & 15 & 16 & 0 \\ 0 & 0 & 17 & 18 & 0 \end{pmatrix} \]
If \( C = AB \), the sum of the diagonal elements of \( C \) is ..............
- IIT JAM CY - 2025
- General Chemistry
A tube fitted with a semipermeable membrane is dipped into 0.001 M NaCl solution at 300 K as shown in the figure. Assume density of the solvent and solution are the same. At equilibrium, the height of the liquid column \( h \) (in cm) is .........

- IIT JAM CY - 2025
- General Chemistry
View More Questions