Explain the following in terms of gain or loss of oxygen with two examples each.
(a) Oxidation
(b) Reduction
(a) Oxidation
(b) Reduction
Approach Solution - 1
(a) Oxidation is the gain of oxygen.
For example: 
In equation (i), H2 is oxidized to H2O and in equation (ii), Cu is oxidised to CuO.
(b) Reduction is the loss of oxygen.
For example : 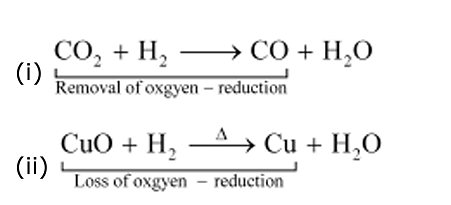
In equation (i), CO2 is reduced to CO and in equation (ii), CuO is reduced to Cu.
Approach Solution -2
In terms of gain or loss of oxygen, oxidation and reduction can be explained as follows:
(a) Oxidation: Oxidation involves the loss of electrons or an increase in oxidation state. When a substance undergoes oxidation, it may also involve the gain of oxygen or the loss of hydrogen.
1. Example of Gain of Oxygen: The oxidation of iron metal to form iron oxide (rust) is a classic example.
\[4Fe + 3O_2 \rightarrow 2Fe_2O_3\]
Here, iron (Fe) is oxidized from an oxidation state of 0 to +3 in iron oxide (Fe2O3), indicating a gain of oxygen.
2. Example of Loss of Oxygen: The combustion of methane (CH4) is an example where methane is oxidized to produce carbon dioxide and water.
\[CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O\]
In this reaction, methane loses hydrogen and gains oxygen, leading to oxidation.
(b) Reduction: Reduction involves the gain of electrons or a decrease in oxidation state. In terms of oxygen, reduction often involves the loss of oxygen or the gain of hydrogen.
1. Example of Loss of Oxygen: The reduction of copper(II) oxide (CuO) using carbon monoxide (CO) to produce copper (Cu) and carbon dioxide (CO2).
\[CuO + CO \rightarrow Cu + CO_2\]
Here, copper(II) oxide loses oxygen to form copper, which is a reduction process.
2. Example of Gain of Oxygen: The oxidation of hydrogen gas (H2) to form water (H2O) is an example where oxygen is gained.
\[2H_2 + O_2 \rightarrow 2H_2O\]
In this reaction, hydrogen gains oxygen, leading to the formation of water, which is an oxidation process for hydrogen.
Top Questions on Effects Of Oxidation Reactions In Everyday Life
- Explain the following terms with one example each.
(a) Corrosion
(b) Rancidity- CBSE Class X
- Science
- Effects Of Oxidation Reactions In Everyday Life
- Oil and fat containing food items are flushed with nitrogen. Why?
- CBSE Class X
- Science
- Effects Of Oxidation Reactions In Everyday Life
- Why do we apply paint on iron articles?
- CBSE Class X
- Science
- Effects Of Oxidation Reactions In Everyday Life
- A shiny brown coloured element ‘X’ on heating in air becomes black in colour. Name the element ‘X’ and the black coloured compound formed.
- CBSE Class X
- Science
- Effects Of Oxidation Reactions In Everyday Life
- What do you mean by a precipitation reaction? Explain by giving examples.
- CBSE Class X
- Science
- Effects Of Oxidation Reactions In Everyday Life
Questions Asked in CBSE X exam
- Find 'mean' and 'mode' of the following data : Frequency Distribution Table
Class 0 – 15 15 – 30 30 – 45 45 – 60 60 – 75 75 – 90 Frequency 11 8 15 7 10 9 - CBSE Class X - 2025
- Statistics
Leaves of the sensitive plant move very quickly in response to ‘touch’. How is this stimulus of touch communicated and explain how the movement takes place?
- CBSE Class X - 2025
- Plant Biology
- Two statements are given below. They are Assertion (A) and Reason (R). Read both the statements carefully and choose the correct option. Assertion (A): Rupees is accepted as medium of exchange in India.
Reason (R): The World Bank legalises the use of rupee as a medium of payment in India.- CBSE Class X - 2025
- Money and Credit
- Two statements are given below. They are Assertion (A) and Reason (R). Read both the statements carefully and choose the correct option:
Assertion (A): Rupees is accepted as medium of exchange in India.
Reason (R): The World Bank legalises the use of rupee as a medium of payment in India.- CBSE Class X - 2025
- Money and Credit
Read the following sources of loan carefully and choose the correct option related to formal sources of credit:
(i) Commercial Bank
(ii) Landlords
(iii) Government
(iv) Money Lende- CBSE Class X - 2025
- Money and Credit
Concepts Used:
Oxidation and Reduction
What is Oxidation?
Oxidation is a chemical process which can be explained by following four point of views
In terms of oxygen transfer
In terms of electron transfer
In terms of hydrogen transfer
In terms of oxidation number
Oxidation in Terms of Oxygen Transfer – Oxidation is gain of oxygen.
What is Reduction?
Reduction is a chemical process which can be explained by following four point of views
In terms of oxygen transfer
In terms of electron transfer
In terms of hydrogen transfer
In terms of oxidation number
Reduction in Terms of Oxygen Transfer – Reduction is loss of oxygen.
What is Redox Reaction?
A chemical reaction which involves transfer of electrons or change in oxidation number of atoms is called redox reaction.