Question:
$28.0\, L$ of $CO _2$ is produced on complete combustion of $168 \, L$ gaseous mixture of ethene and methane at $25^{\circ} C$ and $1 \, atm$ Heat evolved during the combustion process is ______$kJ$ Given : $\Delta H _{ c }\left( CH _4\right)=-900 \, kJ\, mol ^{-1}$ $\Delta H _{ c }\left( C _2 H _4\right)=-1400\, kJ \, mol ^{-1}$
$28.0\, L$ of $CO _2$ is produced on complete combustion of $168 \, L$ gaseous mixture of ethene and methane at $25^{\circ} C$ and $1 \, atm$ Heat evolved during the combustion process is ______$kJ$ Given : $\Delta H _{ c }\left( CH _4\right)=-900 \, kJ\, mol ^{-1}$ $\Delta H _{ c }\left( C _2 H _4\right)=-1400\, kJ \, mol ^{-1}$
Updated On: Feb 3, 2026
Hide Solution
Verified By Collegedunia
Correct Answer: 847 - 848
Approach Solution - 1
The correct answer is 925.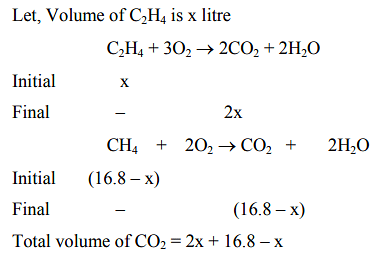
Was this answer helpful?
0
0
Hide Solution
Verified By Collegedunia
Approach Solution -2
The volume of \(\mathrm{CO_2}\) produced enables us to calculate the moles of \(\mathrm{CH_4}\) and \(\mathrm{C_2H_4}\) combusted, from which we can determine the total heat evolved using their respective combustion enthalpies.
Let, the volume of \[ C_2H_4 \text{ be } x \text{ litres.} \]
\[ C_2H_4 + 3O_2 \rightarrow 2CO_2 + 2H_2O \]
Initial volume: \(x\)
Final volume: \(2x\)
For \(\mathrm{CH_4}\), the reaction is \[ CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O \]
Initial volume: \(16.8 - x\)
Final volume: \(16.8 - x\)
The total volume of \(\mathrm{CO_2}\) produced is: \[ \text{Total } CO_2 = 2x + (16.8 - x) \] Thus, \[ 28 = 16.8 + x \] Solving for \(x\), we get \[ x = 11.2 \text{ L} \] Now, calculate the moles of the gases: \[ n_{CH_4} = \frac{PV}{RT} = \frac{1 \times 5.6}{0.082 \times 298} = 0.229 \text{ moles} \] \[ n_{C_2H_4} = \frac{11.2}{0.082 \times 298} = 0.458 \text{ moles} \] Therefore, the heat evolved is: \[ \text{Heat evolved} = 0.229 \times 900 + 0.458 \times 1400 \] \[ = 206.1 + 641.2 = 847.3 \text{ kJ} \]
Let, the volume of \[ C_2H_4 \text{ be } x \text{ litres.} \]
\[ C_2H_4 + 3O_2 \rightarrow 2CO_2 + 2H_2O \]
Initial volume: \(x\)
Final volume: \(2x\)
For \(\mathrm{CH_4}\), the reaction is \[ CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O \]
Initial volume: \(16.8 - x\)
Final volume: \(16.8 - x\)
The total volume of \(\mathrm{CO_2}\) produced is: \[ \text{Total } CO_2 = 2x + (16.8 - x) \] Thus, \[ 28 = 16.8 + x \] Solving for \(x\), we get \[ x = 11.2 \text{ L} \] Now, calculate the moles of the gases: \[ n_{CH_4} = \frac{PV}{RT} = \frac{1 \times 5.6}{0.082 \times 298} = 0.229 \text{ moles} \] \[ n_{C_2H_4} = \frac{11.2}{0.082 \times 298} = 0.458 \text{ moles} \] Therefore, the heat evolved is: \[ \text{Heat evolved} = 0.229 \times 900 + 0.458 \times 1400 \] \[ = 206.1 + 641.2 = 847.3 \text{ kJ} \]
Was this answer helpful?
0
0
Top Questions on Thermodynamics
- 10 mole of an ideal gas is undergoing the process shown in the figure. The heat involved in the process from \( P_1 \) to \( P_2 \) is \( \alpha \) Joule \((P_1 = 21.7 \text{ Pa}, P_2 = 30 \text{ Pa}, C_v = 21 \text{ J/K mol}, R = 8.3 \text{ J/mol K})\). The value of \( \alpha \) is ________.
- JEE Main - 2026
- Physics
- Thermodynamics
- For an ideal gas undergoing an adiabatic process, which quantity remains constant?
- GATE PH - 2026
- Physics
- Thermodynamics
- For an ideal gas, internal energy is a function of:
- In distillation, relative volatility is a measure of:
- Which mode of heat transfer is dominant in vacuum?
View More Questions
Questions Asked in JEE Main exam
Let \( \alpha = \dfrac{-1 + i\sqrt{3}}{2} \) and \( \beta = \dfrac{-1 - i\sqrt{3}}{2} \), where \( i = \sqrt{-1} \). If
\[ (7 - 7\alpha + 9\beta)^{20} + (9 + 7\alpha - 7\beta)^{20} + (-7 + 9\alpha + 7\beta)^{20} + (14 + 7\alpha + 7\beta)^{20} = m^{10}, \] then the value of \( m \) is ___________.- JEE Main - 2026
- Complex Numbers and Quadratic Equations
- The work functions of two metals ($M_A$ and $M_B$) are in the 1 : 2 ratio. When these metals are exposed to photons of energy 6 eV, the kinetic energy of liberated electrons of $M_A$ : $M_B$ is in the ratio of 2.642 : 1. The work functions (in eV) of $M_A$ and $M_B$ are respectively.
- JEE Main - 2026
- Dual nature of matter
- The system of linear equations
$x + y + z = 6$
$2x + 5y + az = 36$
$x + 2y + 3z = b$
has- JEE Main - 2026
- Matrices and Determinants
- The displacement of a particle executing simple harmonic motion with time period \(T\) is expressed as \[ x(t)=A\sin\omega t, \] where \(A\) is the amplitude of oscillation. If the maximum value of the potential energy of the oscillator is found at \[ t=\frac{T}{2\beta}, \] then the value of \(\beta\) is ________.
- JEE Main - 2026
- Waves and Oscillations
- A complex number 'z' satisfy both \(|z-6|=5\) & \(|z+2-6i|=5\) simultaneously. Find the value of \(z^3 + 3z^2 - 15z + 141\).
- JEE Main - 2026
- Algebra
View More Questions