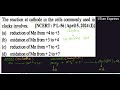
Question:
The reaction at cathode in the cells commonly used in clocks involves.
The reaction at cathode in the cells commonly used in clocks involves.
Updated On: Feb 3, 2026
- reduction of Mn from +4 to +3
- oxidation of Mn from +3 to +4
- reduction of Mn from + 7 to +2
- oxidation of Mn from + 2 to +7
Hide Solution
Verified By Collegedunia
The Correct Option is A
Solution and Explanation
In the cathode reaction manganese (Mn) is reduced from the \(+4\) oxidation state to the \(+3\) state.
Was this answer helpful?
0
2
Learn with videos:
Top Questions on Electrochemistry
- The standard electrode potential of the standard hydrogen electrode (SHE) is:
- GATE CY - 2026
- Chemistry
- Electrochemistry
- For a closed circuit Daniell cell, which of the following plots is the accurate one at a given temperature ?


- JEE Main - 2026
- Chemistry
- Electrochemistry
- Consider the following redox reaction taking place in acidic medium: \[ \mathrm{BH_4^- (aq) + ClO_3^- (aq) \rightarrow H_2BO_3^- (aq) + Cl^- (aq)} \] If the Nernst equation for the above balanced reaction is \[ E_{\text{cell}} = E^\circ_{\text{cell}} - \frac{RT}{nF}\ln Q, \] then the value of \(n\) is ________ (Nearest integer).
- JEE Main - 2026
- Chemistry
- Electrochemistry
- 500 mL of \(1.2\) M KI solution is mixed with 500 mL of \(0.2\) M \( \mathrm{KMnO_4} \) solution in basic medium. The liberated iodine is titrated with standard \(0.1\) M \( \mathrm{Na_2S_2O_3} \) solution in the presence of starch indicator till the blue colour disappears. The volume (in L) of \( \mathrm{Na_2S_2O_3} \) consumed is ________ (nearest integer).
- JEE Main - 2026
- Chemistry
- Electrochemistry
- 'x' is the product from propenenitrile + $SnCl_2/HCl$ followed by hydrolysis. 'y' is the product from but-2-ene by ozonolysis. Which product is not obtained when 'x' and 'y' react in alkali with heating?
- JEE Main - 2026
- Chemistry
- Electrochemistry
View More Questions
Questions Asked in JEE Main exam
- Let \( ABC \) be an equilateral triangle with orthocenter at the origin and the side \( BC \) lying on the line \( x+2\sqrt{2}\,y=4 \). If the coordinates of the vertex \( A \) are \( (\alpha,\beta) \), then the greatest integer less than or equal to \( |\alpha+\sqrt{2}\beta| \) is:
- JEE Main - 2026
- Coordinate Geometry
- Three charges $+2q$, $+3q$ and $-4q$ are situated at $(0,-3a)$, $(2a,0)$ and $(-2a,0)$ respectively in the $x$-$y$ plane. The resultant dipole moment about origin is ___.
- JEE Main - 2026
- Electromagnetic waves
Let \( \alpha = \dfrac{-1 + i\sqrt{3}}{2} \) and \( \beta = \dfrac{-1 - i\sqrt{3}}{2} \), where \( i = \sqrt{-1} \). If
\[ (7 - 7\alpha + 9\beta)^{20} + (9 + 7\alpha - 7\beta)^{20} + (-7 + 9\alpha + 7\beta)^{20} + (14 + 7\alpha + 7\beta)^{20} = m^{10}, \] then the value of \( m \) is ___________.- JEE Main - 2026
- Complex Numbers and Quadratic Equations
- The work functions of two metals ($M_A$ and $M_B$) are in the 1 : 2 ratio. When these metals are exposed to photons of energy 6 eV, the kinetic energy of liberated electrons of $M_A$ : $M_B$ is in the ratio of 2.642 : 1. The work functions (in eV) of $M_A$ and $M_B$ are respectively.
- JEE Main - 2026
- Dual nature of matter
- 10 mole of an ideal gas is undergoing the process shown in the figure. The heat involved in the process from \( P_1 \) to \( P_2 \) is \( \alpha \) Joule \((P_1 = 21.7 \text{ Pa}, P_2 = 30 \text{ Pa}, C_v = 21 \text{ J/K mol}, R = 8.3 \text{ J/mol K})\). The value of \( \alpha \) is ________.
- JEE Main - 2026
- Thermodynamics
View More Questions