Question:
The correct statement(s) regarding P, Q, R, and S is (are) :
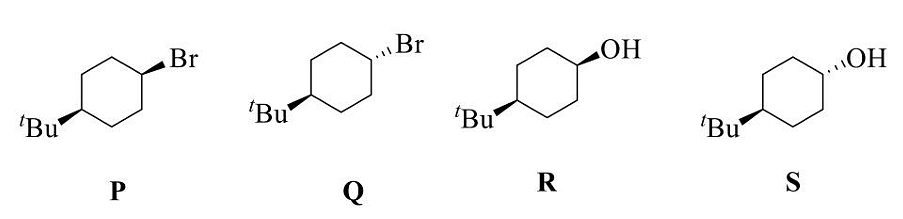
The correct statement(s) regarding P, Q, R, and S is (are) :


Updated On: May 23, 2025
- P reacts faster than Q with PhSNa in DMF as a solvent.
- Q reacts faster than P with NaN3 in DMF as a solvent.
- R reacts faster than S when treated with TSCI/Et3N in DCM as a solvent.
- R gets oxidized faster than S when reacted with CrO3 in DCM as a solvent.
Hide Solution
Verified By Collegedunia
The Correct Option is A, D
Solution and Explanation
The correct option is (A) : P reacts faster than Q with PhSNa in DMF as a solvent and (D) : R gets oxidized faster than S when reacted with CrO3 in DCM as a solvent.
Was this answer helpful?
1
0
Top Questions on Basic mechanistic concepts
- In the given reaction sequence, the amount of R produced (in g) is ______.
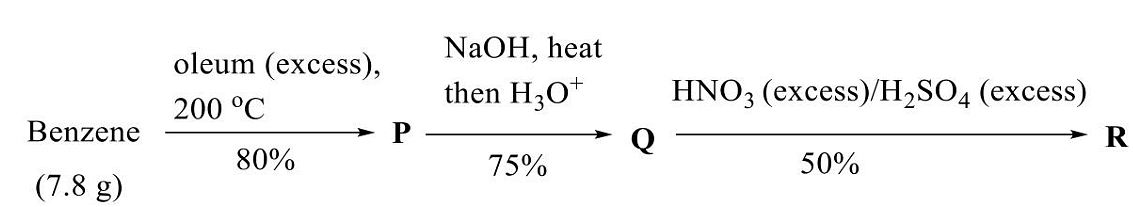
(Given: molar mass (in g mol-1) of H = 1, C = 12, N = 14, O = 16, and S = 32)
(rounded off to two decimal places)- GATE CY - 2024
- Organic Chemistry
- Basic mechanistic concepts
- The correct option(s) that give(s) P as the major product is (are)
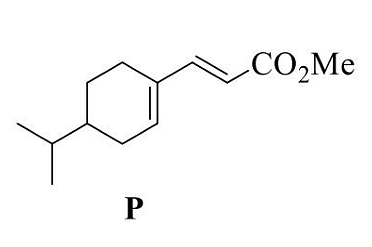
- GATE CY - 2024
- Organic Chemistry
- Basic mechanistic concepts
- The major product in the following reaction sequence is
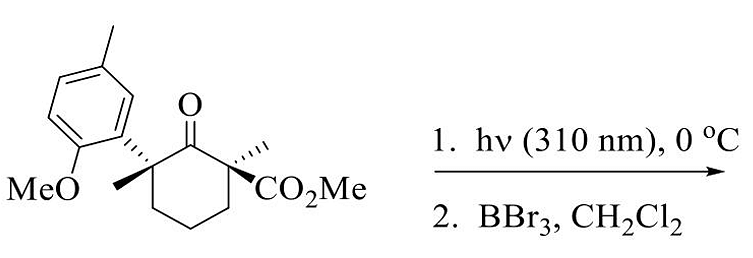
- GATE CY - 2024
- Organic Chemistry
- Basic mechanistic concepts
Questions Asked in GATE CY exam
- For a first-order reaction, the unit of rate constant is:
- GATE CY - 2026
- Chemical Kinetics
- Which ligand causes maximum crystal field splitting?
- GATE CY - 2026
- Coordination chemistry
- The standard electrode potential of the standard hydrogen electrode (SHE) is:
- GATE CY - 2026
- Electrochemistry
- Which of the following reagents converts an aldehyde selectively into a primary alcohol?
- GATE CY - 2026
- Organic Chemistry
- In IR spectroscopy, which bond absorbs at the highest wavenumber?
- GATE CY - 2026
- Spectroscopy
View More Questions