Question:
Phenol on reduction with H2 in the presence of Ni catalyst gives:
Phenol on reduction with H2 in the presence of Ni catalyst gives:
Updated On: Sep 30, 2024
- (A) Benzene
- (B) Toulene
- (C) Cyclohexane
(D) Cyclohexanol
Hide Solution
Verified By Collegedunia
The Correct Option is D
Solution and Explanation
Explanation:
Phenol is hydrogenated to cyclohexanol,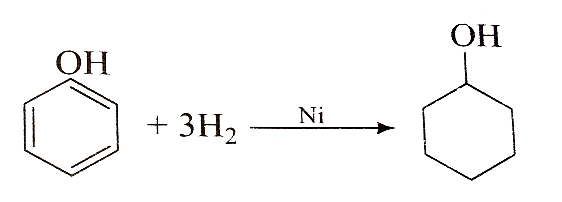
Phenol is hydrogenated to cyclohexanol,
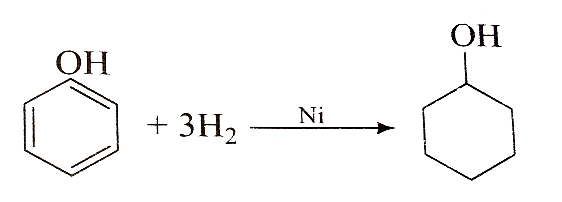
Was this answer helpful?
0
1
Top Questions on Alcohols, Phenols and Ethers
From the following, how many compounds contain at least one secondary alcohol?

- JEE Main - 2026
- Chemistry
- Alcohols, Phenols and Ethers
- Organic compound (P) $\xrightarrow{(i) \text{ excess of} HI, (ii) \text{ Aq. } NaOH} Q + R$. $Q$ and $R$ both gives Iodoform test, Which among the following is (P) from the given organic compound?

- JEE Main - 2026
- Chemistry
- Alcohols, Phenols and Ethers
Given below are two statements:
Statement I: Dimethyl ether is completely soluble in water. However, diethyl ether is soluble in water to a very small extent.
Statement II: Sodium metal can be used to dry diethyl ether and not ethyl alcohol.
In the light of the given statements, choose the correct answer from the options given below:- JEE Main - 2025
- Chemistry
- Alcohols, Phenols and Ethers
- Why is ethyl alcohol not dried by anhydrous CaCl2?
- Bihar Board XII - 2025
- Chemistry
- Alcohols, Phenols and Ethers
- An alcohol X (C$_4$H$_{10}$O) reacts with Conc.HCl/ZnCl$_2$ to give corresponding chloride. X on dehydration forms Y, which reacts with Baeyer's reagent to give Z. What is Z?
- TS EAMCET - 2025
- Chemistry
- Alcohols, Phenols and Ethers
View More Questions
Questions Asked in JEE Main exam
- A 20 m long uniform copper wire held horizontally is allowed to fall under the gravity (g = 10 m/s²) through a uniform horizontal magnetic field of 0.5 Gauss perpendicular to the length of the wire. The induced EMF across the wire when it travels a vertical distance of 200 m is_______ mV.}
- JEE Main - 2026
- Thermodynamics
- If the end points of chord of parabola \(y^2 = 12x\) are \((x_1, y_1)\) and \((x_2, y_2)\) and it subtend \(90^\circ\) at the vertex of parabola then \((x_1x_2 - y_1y_2)\) equals :
- JEE Main - 2026
- Probability
- The sum of all possible values of \( n \in \mathbb{N} \), so that the coefficients of \(x, x^2\) and \(x^3\) in the expansion of \((1+x^2)^2(1+x)^n\) are in arithmetic progression is :
- JEE Main - 2026
- Integration
- In a microscope of tube length $10\,\text{cm}$ two convex lenses are arranged with focal lengths $2\,\text{cm}$ and $5\,\text{cm}$. Total magnification obtained with this system for normal adjustment is $(5)^k$. The value of $k$ is ___.
- JEE Main - 2026
- Optical Instruments
Which one of the following graphs accurately represents the plot of partial pressure of CS₂ vs its mole fraction in a mixture of acetone and CS₂ at constant temperature?


- JEE Main - 2026
- Organic Chemistry
View More Questions