In the structure of \({ClF_3}\), the number of lone pairs of electrons on the central atom \(Cl\) is:
- three
- one
- four
- two
The Correct Option is D
Approach Solution - 1
The correct answer is Option D) two
The structure of \(ClF _{3}\) is 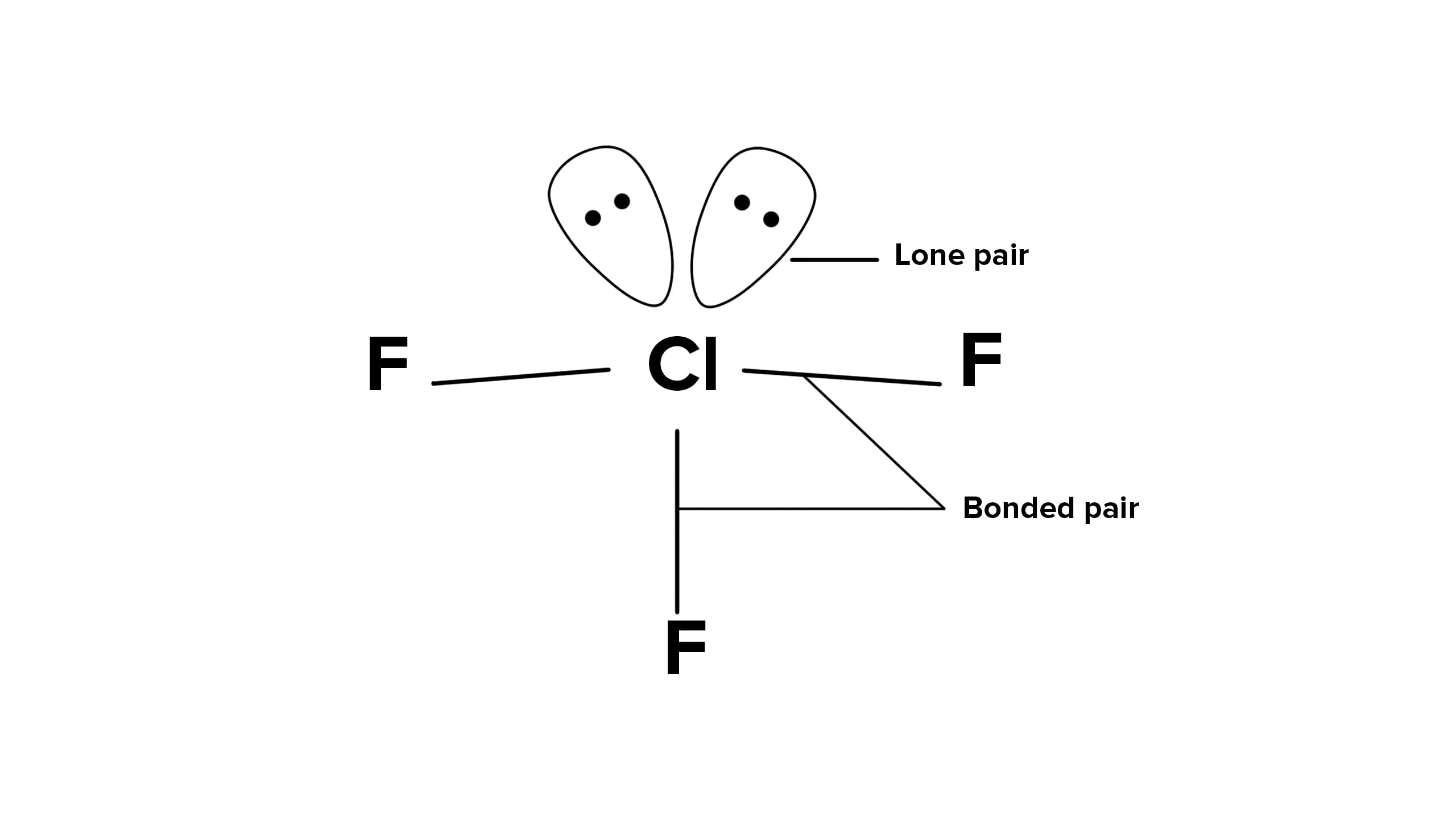
In the structure of ClF3, the number of lone pairs of electrons on the central atom Cl is two. Cl has 7 valence electrons out of which 3 are involved in bond formation with 3 F atoms. 7−3=4 valence electrons remain in the form of 2 lone pairs of electrons.
Approach Solution -2
The correct answer is Option D) two
Some real-life examples of p-block elements:
1. Carbon: It is the building block of all organic compounds. Carbon is also used in many other applications, such as making plastics, steel, and semiconductors.
2. Silicon: It is used to make semiconductors and also used in making solar cells and other renewable energy technologies.
3. Phosphorus: It is also used in making matches, fertilizers, and fireworks.
4. Germanium: It is used in making transistors and other electronic devices, and also used in making infrared detectors and other optical devices.
Question can also be asked as
1. How many lone pairs of electrons does the chlorine atom in ClF3 have?
2. What is the number of unshared pairs of electrons on the chlorine atom in ClF3?
3. How many bonding pairs of electrons does the chlorine atom in ClF3 have?
Learn with videos:
Top Questions on Chemical bonding and molecular structure
From the given following (A to D) cyclic structures, those which will not react with Tollen's reagent are :

- JEE Main - 2026
- Chemistry
- Chemical bonding and molecular structure
- The wave numbers of three spectral lines of hydrogen atom are considered. Identify the set of spectral lines belonging to the {Balmer series. (\(R\) = Rydberg constant)}
- JEE Main - 2026
- Chemistry
- Chemical bonding and molecular structure
- Consider the reaction: \[ \text{Ph–CH=CH}_2 \xrightarrow[\text{peroxide}]{\text{HBr}} \text{Product} \] Which of the following statements are correct?
[A.] The reaction proceeds through a more stable radical intermediate.
[B.] The role of peroxide is to generate \(\mathrm{H^\bullet}\) radical.
[C.] During this reaction, benzene is formed as a byproduct.
[D.] \(1\)-Bromo-\(2\)-phenylethane is formed as a minor product.
[E.] The same reaction in absence of peroxide proceeds via a carbocation intermediate. Choose the correct answer.- JEE Main - 2026
- Chemistry
- Chemical bonding and molecular structure
Compound 'P' undergoes the following sequence of reactions : (i) NH₃ (ii) $\Delta$ $\rightarrow$ Q (i) KOH, Br₂ (ii) CHCl₃, KOH (alc), $\Delta$ $\rightarrow$ NC-CH₃. 'P' is :


- JEE Main - 2026
- Chemistry
- Chemical bonding and molecular structure
- Given below are two statements :
Statement I : The correct order in terms of bond dissociation enthalpy is \( Cl_2>Br_2>F_2>I_2 \).
Statement II : The correct trend in the covalent character of the metal halides is \( SnCl_2>SnCl_4 \), \( PbCl_2>PbCl_4 \) and \( UF_4>UF_6 \).
In the light of the above statements, choose the correct answer from the options given below :- JEE Main - 2026
- Chemistry
- Chemical bonding and molecular structure
Questions Asked in NEET exam
- Two cities X and Y are connected by a regular bus service with a bus leaving in either direction every T min. A girl is driving scooty with a speed of 60 km/h in the direction X to Y. She notices that a bus goes past her every 30 minutes in the direction of her motion, and every 10 minutes in the opposite direction. Choose the correct option for the period T of the bus service and the speed (assumed constant) of the buses.
- NEET (UG) - 2025
- Relative Velocity
- A physical quantity P is related to four observations a, b, c, and d as follows: P = a3 b2 (c / √d) The percentage errors of measurement in a, b, c, and d are 1%, 3%, 2%, and 4% respectively. The percentage error in the quantity P is:
- NEET (UG) - 2025
- Dimensional analysis and its applications
What is Microalbuminuria ?
- NEET (UG) - 2025
- Human physiology
The output (Y) of the given logic implementation is similar to the output of an/a …………. gate.

- NEET (UG) - 2025
- Logic gates
- An oxygen cylinder of volume 30 litre has 18.20 moles of oxygen. After some oxygen is withdrawn from the cylinder, its gauge pressure drops to 11 atmospheric pressure at temperature \(27^\circ\)C. The mass of the oxygen withdrawn from the cylinder is nearly equal to: [Given, \(R = \frac{100}{12} \text{ J mol}^{-1} \text{K}^{-1}\), and molecular mass of \(O_2 = 32 \text{ g/mol}\), 1 atm pressure = \(1.01 \times 10^5 \text{ N/m}^2\)]
- NEET (UG) - 2025
- Ideal-gas equation and absolute temperature
Concepts Used:
Chemical Bonding and Molecular Structure
Such a group of atoms is called a molecule. Obviously, there must be some force that holds these constituent atoms together in the molecules. The attractive force which holds various constituents (atoms, ions, etc.) together in different chemical species is called a chemical bond.
Types of Chemical Bonds:
There are 4 types of chemical bonds which are formed by atoms or molecules to yield compounds.
- Ionic Bonds - Ionic bonding is a type of chemical bonding which involves a transfer of electrons from one atom or molecule to another.
- Covalent Bonds - Compounds that contain carbon commonly exhibit this type of chemical bonding.
- Hydrogen Bonds - It is a type of polar covalent bonding between oxygen and hydrogen wherein the hydrogen develops a partial positive charge
- Polar Bonds - In Polar Covalent chemical bonding, electrons are shared unequally since the more electronegative atom pulls the electron pair closer to itself and away from the less electronegative atom.
Factors Affecting Bond Enthalpy in Chemical Bonding:
- Size of the Atom
- Multiplicity of Bonds
- Number of Lone Pair of Electrons Present
- Bond Angle