Question:
Electrometallurgical process is used to extract
Electrometallurgical process is used to extract
Updated On: Jun 18, 2022
- Fe
- Pb
- Na
- Ag
Hide Solution
Verified By Collegedunia
The Correct Option is C
Solution and Explanation
Because $Na$ is very reactive and cannot be extracted by means of the reduction by $C , CO$ etc.
So extracted by electrolysis.
So extracted by electrolysis.
Was this answer helpful?
0
0
Top Questions on General Principles and Processes of Isolation of Elements
- What is used for the Thermite Reaction?
- BCECE - 2025
- Chemistry
- General Principles and Processes of Isolation of Elements
- In the extraction of iron using blast furnace to remove the impurity (X), chemical (Y) is added to the ore. X and Y are respectively
- TS EAMCET - 2025
- Chemistry
- General Principles and Processes of Isolation of Elements
- Which of the following compounds is used to cover the surface of a metallic object to prevent corrosion?
- KEAM - 2025
- Chemistry
- General Principles and Processes of Isolation of Elements
- The incorrect statement about the Hall-Heroult process is:
- KCET - 2024
- Chemistry
- General Principles and Processes of Isolation of Elements
- Select the correct statement:
- KCET - 2024
- Chemistry
- General Principles and Processes of Isolation of Elements
View More Questions
Questions Asked in BITSAT exam
- What is the dot product of the vectors \( \mathbf{a} = (2, 3, 1) \) and \( \mathbf{b} = (1, -1, 4) \)?
- BITSAT - 2025
- Vector Algebra
- Find the determinant of the matrix \( A = \begin{bmatrix} 2 & 3 \\ 4 & 5 \end{bmatrix} \).
- BITSAT - 2025
- Matrices
- A convex lens has focal length 20 cm. An object is placed at a distance of 40 cm from the lens. What is the position of the image formed?
- BITSAT - 2025
- Ray optics and optical instruments
- What is the value of \( \sin 30^\circ \)?
- BITSAT - 2025
- Trigonometry
- The area enclosed between the curve \(y = \log_e(x + e)\) and the coordinate axes is:
- BITSAT - 2025
- Fundamental Theorem of Calculus
View More Questions
Concepts Used:
General Principles and Processes of Isolation of Elements
What are Ores and Minerals?
Minerals are the naturally occurring, homogeneous inorganic solid substances. They are having a definite chemical composition and crystalline structure, hardness and color. For example, copper pyrite, calamine, etc.
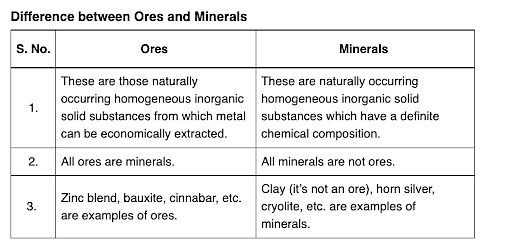
Impurities in an ore are called gauge. The removal of a gauge from the ore is called concentration ore.
Several steps are involved in the extraction of pure metal from ores. Major steps are as follows –
- Concentration of the ore
- Isolation of the metal from its concentrated ore
- Purification of the metal