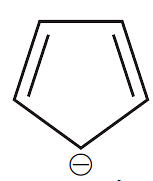
Cyclopentadienyl anion is
- benzenoid and aromatic
- non-benzenoid and aromatic
- non-benzenoid and non-aromatic
- non-benzenoid and anti-aromatic
The Correct Option is B
Solution and Explanation
planar with $(4 n+2) \pi e^{-}$. Therefore
aromatic but structure is not similar to benzene.
Top Questions on Aromatic Hydrocarbon
Which of the following are aromatic?

- CUET (PG) - 2025
- Chemistry
- Aromatic Hydrocarbon
- When C6H5CHO reacts with the mixture of HNO3 and H2SO4 at 273-283K, it gives
- KEAM - 2025
- Chemistry
- Aromatic Hydrocarbon
- The temperature and pressure required for reforming benzene from n-hexane is
- KEAM - 2025
- Chemistry
- Aromatic Hydrocarbon
- Which of the following is non-aromatic?
- KEAM - 2025
- Chemistry
- Aromatic Hydrocarbon
Give plausible explanation for:
(a) Diazonium salts of aromatic amines are stable.
(b) Aniline does not undergo Friedel-Crafts reaction.
(c) Aniline on nitration gives substantial meta product.- CBSE CLASS XII - 2024
- Chemistry
- Aromatic Hydrocarbon
Questions Asked in TS EAMCET exam
- A body is projected vertically upward with an initial velocity of 40 m/s. Calculate the maximum height reached by the body. (Take \( g = 9.8 \, \text{m/s}^2 \))
- TS EAMCET - 2025
- Kinematics
- The properties required for a material to be used as the core of an electromagnet are:
- TS EAMCET - 2025
- Magnetism and matter
- Which of the following statements are correct?
- TS EAMCET - 2025
- Colligative Properties
- At 27$^\circ$C, 100 mL of 0.05 M Cu$^{2+}$ solution is added to 1 L of 0.1 M KI. Find [KI] in resultant solution.
- TS EAMCET - 2025
- Stoichiometry and Stoichiometric Calculations
- 4.0 g of a mixture containing Na₂CO₃ and NaHCO₃ is heated to 673K. Loss in mass of the mixture is found to be 0.62g. The percentage of sodium carbonate in the mixture is
- TS EAMCET - 2025
- Stoichiometry and Stoichiometric Calculations
Concepts Used:
Aromatic hydrocarbon
Aromatic hydrocarbons, sometimes known as arenes, are aromatic organic molecules made up entirely of carbon and hydrogen. In aromatic compounds a benzene ring which is named after the simple aromatic chemical benzene, or a phenyl group when part of a larger structure, is the configuration of six carbon atoms.
Read More: Aromaticity
Reactions of Aromatic Hydrocarbons:
1. Aromatic Substitution Reactions
This reaction involves the replacement of one substituent on the ring of an aromatic hydrocarbon, commonly a hydrogen atom, by a different substituent group.
The common types of aromatic substitution reactions are:
- Nucleophilic aromatic substitution reactions
- Electrophilic aromatic substitution reactions
- Radical nucleophilic aromatic substitution reactions
2. Coupling Reactions
In these types of reactions, the coupling of two fragments that have a radical nature is achieved with the help of a metal catalyst