Question:
According to the SAR of Chloroquine, electron:
According to the SAR of Chloroquine, electron:
Show Hint
- Chloroquine’s antimalarial activity depends on its ability to inhibit hemozoin formation.
- Electron-withdrawing groups at the 7th position enhance this inhibition.
- Electron-donating groups decrease efficacy by altering drug-target interactions.
Updated On: Feb 4, 2025
- Withdrawing group at 6th position of the quinoline ring is important for the inhibition of hemozoin formation
- Donating group at 7th position of the quinoline ring is important for the inhibition of hemozoin formation
- Donating group at 6th position of the quinoline ring is important for the inhibition of hemozoin formation
- Withdrawing group at 7th position of the quinoline ring is important for the inhibition of hemozoin formation
Hide Solution
Verified By Collegedunia
The Correct Option is D
Solution and Explanation
The Structure-Activity Relationship (SAR) of Chloroquine indicates that the quinoline ring plays a crucial role in the drug's ability to inhibit hemozoin formation, which is essential for its antimalarial activity. The presence of an electron-withdrawing group at the 7th position of the quinoline ring enhances the drug’s potency by stabilizing interactions with the target site and inhibiting the polymerization of heme into hemozoin.
Was this answer helpful?
0
0
Top Questions on Phytopharmaceuticals
- According to the SAR of Chloroquine electron:
- GPAT - 2024
- Pharmacognosy
- Phytopharmaceuticals
- Following is not an example of carbapenem
- GPAT - 2023
- Pharmacognosy
- Phytopharmaceuticals
- Quinine and quinidine differs in:
- GPAT - 2023
- Pharmacognosy
- Phytopharmaceuticals
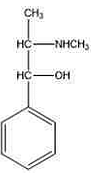
The above structure is of:- GPAT - 2022
- Pharmacognosy
- Phytopharmaceuticals
Rancidity in the fixed oils generally show ‐
- GPAT - 2022
- Pharmacognosy
- Phytopharmaceuticals
View More Questions
Questions Asked in GPAT exam
- Match the following:
(1) Schedule FF
(2) Schedule F3
(3) Schedule V
(4) Schedule Y
Descriptions:
(P) Standards of patent and proprietary medicines
(Q) Requirements and guidelines for clinical trials
(R) Standards for sterilized umbilical tapes
(S) Standards for Ophthalmic preparations- GPAT - 2025
- Drug therapy
- If the label or the container bears the name of an individual or company purporting to be the manufacturer of the drug, which individual or company is fictitious or does not exist, it is:
- GPAT - 2025
- Drug therapy
- Manufacturing Specification for tooling has been standardized by?
- GPAT - 2025
- Pharmacy Profession & Introduction to Pharmaceuticals
- As per USP, the maximum concentration of benzalkonium chloride used as a preservative in parenteral formulations is
- GPAT - 2025
- Pharmaceutical Analysis
Match the following:
(P) Schedule H
(Q) Schedule G
(R) Schedule P
(S) Schedule F2
Descriptions:
(I) Life period of drugs
(II) Drugs used under RMP
(III) List of Prescription Drugs
(IV) Standards for surgical dressing
- GPAT - 2025
- Drug therapy
View More Questions