Question:
Which one of the following metals is extracted by a carbon reduction process?
Which one of the following metals is extracted by a carbon reduction process?
Updated On: Jun 8, 2024
- Copper
- Iron
- Aluminium
- Magnesium
Hide Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
The less electropositive metals such as $Fe,\, Zn,\, Sn$ etc are extracted from their oxides by reduction with carbon or coal.
$Fe _{2} O _{3}+3 C \rightarrow 2 Fe +3 CO$
$Fe _{2} O _{3}+3 CO \rightarrow \underset{\text { iron}}{2 Fe }+3 CO _{2}$
$Fe _{2} O _{3}+3 C \rightarrow 2 Fe +3 CO$
$Fe _{2} O _{3}+3 CO \rightarrow \underset{\text { iron}}{2 Fe }+3 CO _{2}$
Was this answer helpful?
0
0
Top Questions on General Principles and Processes of Isolation of Elements
- What is used for the Thermite Reaction?
- BCECE - 2025
- Chemistry
- General Principles and Processes of Isolation of Elements
- In the extraction of iron using blast furnace to remove the impurity (X), chemical (Y) is added to the ore. X and Y are respectively
- TS EAMCET - 2025
- Chemistry
- General Principles and Processes of Isolation of Elements
- Which of the following compounds is used to cover the surface of a metallic object to prevent corrosion?
- KEAM - 2025
- Chemistry
- General Principles and Processes of Isolation of Elements
- The incorrect statement about the Hall-Heroult process is:
- KCET - 2024
- Chemistry
- General Principles and Processes of Isolation of Elements
- Select the correct statement:
- KCET - 2024
- Chemistry
- General Principles and Processes of Isolation of Elements
View More Questions
Questions Asked in JKCET exam
- Which of the following is not an example of an ideal solution?
- JKCET - 2024
- Colligative Properties
- What is the value of the Van't Hoff factor (i) for solutes that dissociate in water?
- JKCET - 2024
- Colligative Properties
- Which of the following do not show geometrical isomerism? (Assume all ligands are unidentate)
- JKCET - 2024
- coordination compounds
- Lewis concept does explain the behaviour of
- JKCET - 2024
- Acids and Bases
- In the reaction, $ H_2(g) + Br_2(g) = 2HBr(g) $, what will happen if there is a change in pressure?
- JKCET - 2024
- Law Of Chemical Equilibrium And Equilibrium Constant
View More Questions
Concepts Used:
General Principles and Processes of Isolation of Elements
What are Ores and Minerals?
Minerals are the naturally occurring, homogeneous inorganic solid substances. They are having a definite chemical composition and crystalline structure, hardness and color. For example, copper pyrite, calamine, etc.
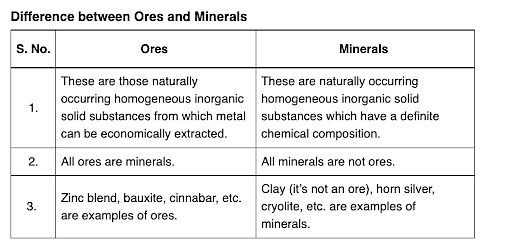
Impurities in an ore are called gauge. The removal of a gauge from the ore is called concentration ore.
Several steps are involved in the extraction of pure metal from ores. Major steps are as follows –
- Concentration of the ore
- Isolation of the metal from its concentrated ore
- Purification of the metal